IVF SUPPORT
Science-backed red light laser therapy designed to support circulation, uterine health, cellular energy and implantation readiness throughout IVF and assisted reproductive treatment.
Designed to work alongside your fertility clinic and treatment protocol.

Who This May Support
Women navigating:
- IVF, ICSI or embryo transfer preparation
- recurrent implantation challenges
- frozen embryo transfer or donor egg cycles
- IVF-related stress and overwhelm
- uterine lining and circulation support
- a desire for gentle, non-invasive care alongside fertility treatment

IVF can feel physically and emotionally consuming.
Many women come to Luma looking for ways to feel more proactive, supported and connected to their body throughout treatment.
Our approach is designed to work alongside your fertility clinic — not replace it — while helping support your body through each stage of the IVF process.
How Red Light Therapy May Support IVF and Assisted Reproductive Treatment
Red light therapy — also known as photobiomodulation or low-level laser therapy (LLLT) — is being explored as a supportive therapy alongside assisted reproductive treatment, including IVF, donor egg IVF, ICSI and frozen embryo transfer cycles.
Research in this area is still evolving. While photobiomodulation is not part of standard IVF protocols, emerging studies and clinical observations suggest it may help support circulation, cellular energy, uterine receptivity and reproductive tissue function.
Implantation Support and Uterine Receptivity
Successful IVF depends on both embryo quality and endometrial receptivity at the time of embryo transfer. Reduced uterine blood flow, impaired endometrial development, and altered cellular metabolism are recognised contributors to implantation failure.
A single-blinded randomised clinical trial in women with recurrent implantation failure undergoing IVF investigated transabdominal photobiomodulation and reported higher biochemical and clinical pregnancy rates in the treatment group compared with controls, although results were limited by small sample size.
The authors concluded that photobiomodulation demonstrated biological plausibility and supported the need for larger, well-designed IVF studies.
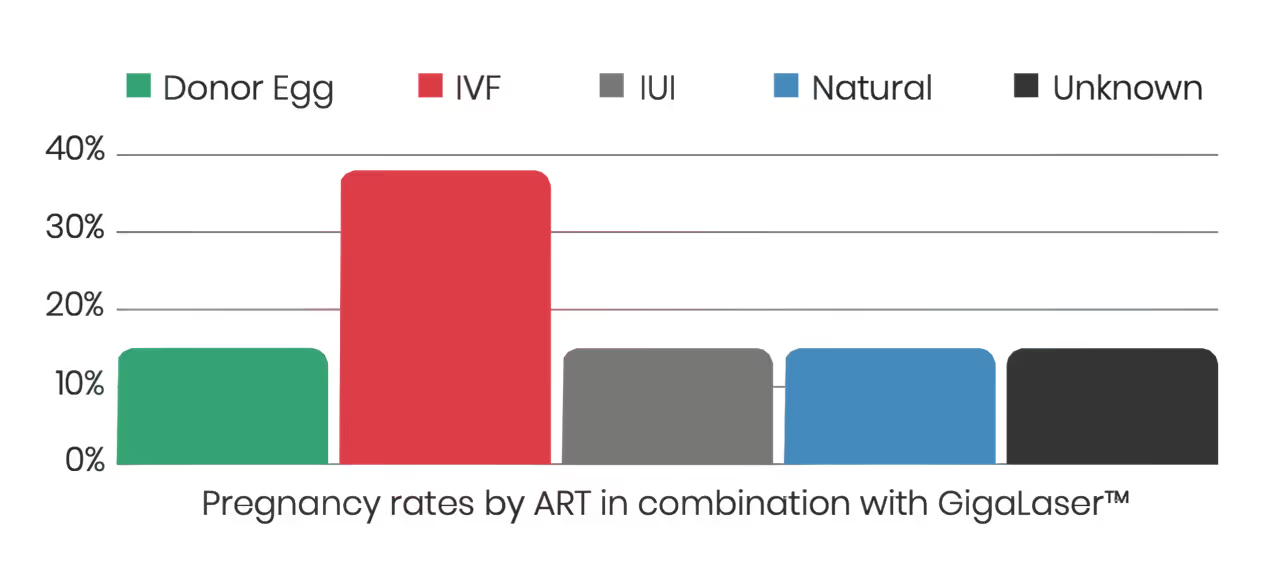
Clinical Case Series Using the GigaLaser with Anne-Marie Jensen
A published clinical case series led by Anne-Marie Jensen examined the use of structured red and near-infrared photobiomodulation protocols in women with complex or treatment-resistant infertility.
In this series, photobiomodulation was delivered using high-coverage laser systems, including the GigaLaser, which is designed to deliver red and near-infrared wavelengths across the pelvic region.
The reported cases involved women with long-standing infertility histories and previous ART failures. Following the integration of photobiomodulation alongside conventional fertility care, the series documented clinical pregnancies and live births.
Donor Egg IVF and the Endometrial Environment
In donor egg IVF, embryo quality is typically high, and implantation success is largely dependent on uterine receptivity. Thin endometrium, impaired blood flow, and altered endometrial metabolism are common clinical challenges.
Early observational fertility studies using multi-wavelength red and near-infrared photobiomodulation have reported improved reproductive outcomes in women with previous implantation failure despite good-quality embryos.
While these findings are preliminary, they suggest that photobiomodulation may influence uterine physiology in ways that support implantation, warranting further controlled investigation.
Sperm Motility and Function
In vitro research has shown that red light laser exposure can improve sperm motility and metabolic activity.
A controlled laboratory study demonstrated that exposure to 650 nm red laser light significantly increased progressive sperm motility compared with untreated samples.
A systematic review examining photobiomodulation in male infertility reported consistent improvements in sperm motility and mitochondrial activity across multiple studies.
These findings suggest a potential supportive role for photobiomodulation in relation to sperm quality, although effects on clinical pregnancy outcomes following IUI remain to be established.

Biological Mechanisms Relevant to Reproductive Tissue
The rationale for photobiomodulation in reproductive health is supported by a substantial body of basic science research.
Red and near-infrared light has been shown to stimulate mitochondrial respiratory chain activity, increase ATP production, and modulate reactive oxygen species signalling, all of which are fundamental to cellular energy and viability.
Additional studies have demonstrated improvements in microcirculation and tissue oxygenation following photobiomodulation, providing a plausible physiological basis for its investigation in reproductive tissues.
Experimental models have also shown reduced oxidative stress and improved ovarian function following photobiomodulation exposure.
Research and Clinical References
Emerging research continues to explore how photobiomodulation may support circulation, cellular repair and reproductive function alongside assisted reproductive treatment.
- Khorsandi et al.
Effect of photobiomodulation therapy on recurrent implantation failure in IVF patients: a randomised clinical trial.
Journal of Clinical Medicine. 2024.
https://pubmed.ncbi.nlm.nih.gov/39051002/ - Hanna, et al.
Multi-wavelength photobiomodulation therapy in female infertility: a clinical case series with live births.
Journal of Clinical Medicine. 2024.
https://www.mdpi.com/2077-0383/13/23/7101 - Alves et al.
Effect of low-level red laser irradiation on human sperm motility: an in vitro study.
Lasers in Medical Science. 2023.
https://pubmed.ncbi.nlm.nih.gov/36610824/ - Hamblin MR, et al.
Photobiomodulation and male infertility: a systematic review.
Andrology. 2023.
https://pubmed.ncbi.nlm.nih.gov/38028870/ - de Freitas LF, Hamblin MR.
Mechanisms of photobiomodulation: cellular and mitochondrial responses.
Lasers in Medical Science.
https://pubmed.ncbi.nlm.nih.gov/39095677/ - Chung H, et al.
The nuts and bolts of low-level laser (light) therapy.
Annals of Biomedical Engineering.
https://pubmed.ncbi.nlm.nih.gov/37041786/ - Silveira et al.
Photobiomodulation improves ovarian function by reducing oxidative stress in experimental models.
Lasers in Medical Science.
https://www.sciencedirect.com/science/article/abs/pii/S1011134424001842








